6.2 Thermal Energy
How can containers keep stuff from warming up or cooling down?

Unit Summary
What keeps different cups or containers from warming up or cooling down? Students begin this 6th grade science unit by experimenting whether a new plastic cup sold by a store keeps a drink colder for longer than the regular plastic cup that comes free with the drink. Students find that the drink in the regular cup warms up more than the drink in the special cup. This prompts students to identify features of the cups that are different, such as the lid, walls, and hole for the straw, that might explain why one drink warms up more than the other.
In this 6th grade science unit, students investigate the different cup features they conjecture to explain the phenomenon, starting with the lid. They model how matter can enter or exit the cup via evaporation. However, they find that in a completely closed system, the liquid inside the cup still changes temperature. This motivates the need to trace the transfer of energy into the drink as it warms up. Through a series of lab investigations and simulations, students find two ways to transfer energy into the drink: (1) the absorption of light and (2) thermal energy from the warmer air around the drink. They are then challenged to design their own drink container that can perform as well as the store-bought container, following a set of design criteria and constraints.

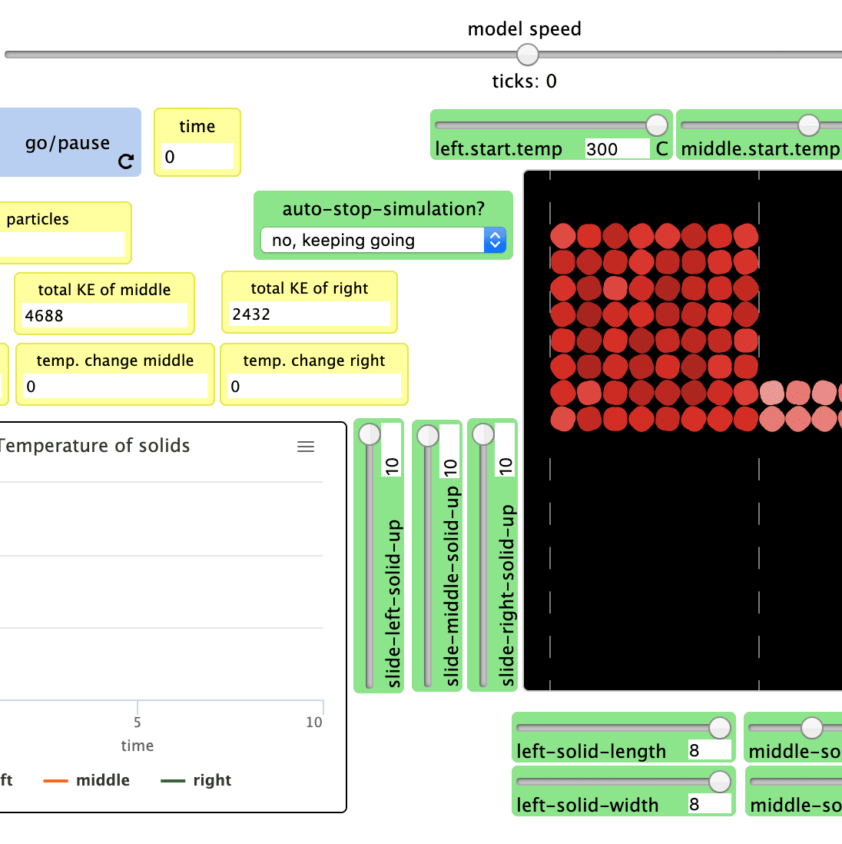
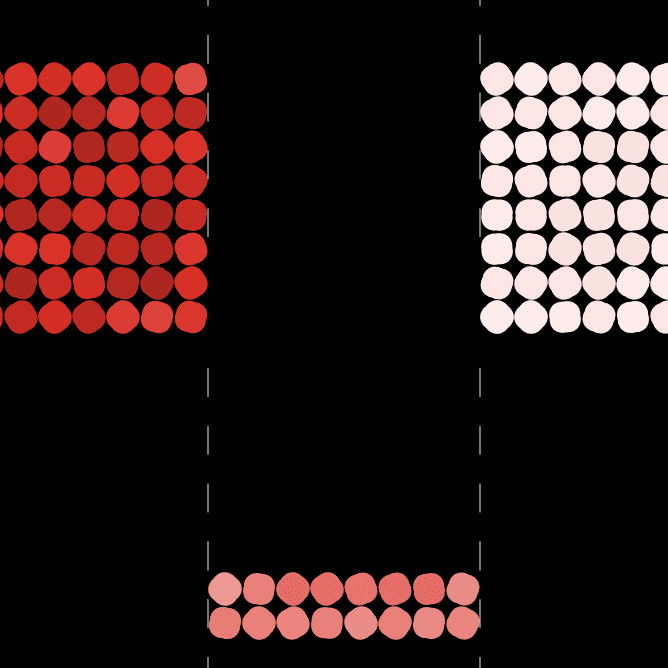
Simulations
Unit Examples
Additional Unit Information
Next Generation Science Standards Addressed in this Unit
Performance Expectations

This 6th grade science unit on thermal energy builds toward the following NGSS Performance Expectations (PEs):
- MS-PS1-4*: Develop a model that predicts and describes changes in particle motion, temperature, and state of a pure substance when thermal energy is added or removed.
- MS-PS3-3: Apply scientific principles to design, construct, and test a device that either minimizes or maximizes thermal energy transfer
- MS-PS3-4: Plan an investigation to determine the relationships among the energy transferred, the type of matter, the mass, and the change in the average kinetic energy of the particles as measured by the temperature of the sample
- MS-PS3-5: Construct, use, and present arguments to support the claim that when the kinetic energy of an object changes, energy is transferred to or from the object.
- MS-PS4-2*: Develop and use a model to describe that waves are reflected, absorbed, or transmitted through various materials.
- MS-ETS1-4: Develop a model to generate data for iterative testing and modification of a proposed object, tool, or process such that an optimal design can be achieved.
*These performance expectations are developed across multiple units. This unit reinforces or works toward these NGSS PEs that students should have previously developed or will develop more fully in future units. In the OpenSciEd Scope and Sequence, PS4-2 is first built in Unit 6.1. In this new context of particle models and energy transfer, students learn more about how absorption of light occurs at the particle level. This unit begins to address changes in state that are part of PS1-4; then changes in state are more fully developed in the next unit, 6.3, on water cycling. In 6.3, students learn that evaporation and condensation occur when energy is added or removed from the substance.
Disciplinary Core Ideas

The 6th grade science unit on thermal energy expands students’ understanding of particle models and energy transfer, which include these Grade 6–8 DCI elements:
PS1.A: Structure and Properties of Matter
- Gases and liquids are made of molecules or inert atoms that are moving about relative to each other. (MS‑PS1‑4)
- In a liquid, the molecules are constantly in contact with others; in a gas, they are widely spaced except when they happen to collide. In a solid, atoms are closely spaced and may vibrate in position but do not change relative locations. (MS‑PS1‑4)
- The changes of state that occur with variations in temperature or pressure can be described and predicted using these models of matter. (MS‑PS1‑4)
PS3.A: Definitions of Energy
- The term “heat” as used in everyday language refers both to thermal energy (the motion of atoms or molecules within a substance) and the transfer of that thermal energy from one object to another. In science, heat is used only for this second meaning; it refers to the energy transferred due to the temperature difference between two objects. (secondary to MS‑PS1‑4)
- Temperature is not a measure of energy; the relationship between the temperature and the total energy of a system depends on the types, states, and amounts of matter present. (secondary to MS‑PS1‑4)
- Temperature is a measure of the average kinetic energy of particles of matter. The relationship between the temperature and the total energy of a system depends on the types, states, and amounts of matter present. (MS‑PS3‑3), (MS‑PS3‑4)
PS3.B: Conservation of Energy and Energy Transfer
- When the kinetic energy of an object changes, there is inevitably some other change in energy at the same time. (MS‑PS3‑5)
- The amount of energy transfer needed to change the temperature of a matter sample by a given amount depends on the nature of the matter, the size of the sample, and the environment. (MS‑PS3‑4)
- Energy is spontaneously transferred out of hotter regions or objects and into colder ones. (MS‑PS3‑3)
PS4.B: Electromagnetic Radiation
- When light shines on an object, it is reflected, absorbed, or transmitted through the object, depending on the object’s material and the frequency (color) of the light. (MS‑PS4‑2)
ETS1.A: Defining and Delimiting an Engineering Problem
- The more precisely a design task’s criteria and constraints can be defined, the more likely it is that the designed solution will be successful. Specification of constraints includes consideration of scientific principles and other relevant knowledge that is likely to limit possible solutions. (secondary to MS‑PS3‑3)
ETS1.B: Developing Possible Solutions
- A solution needs to be tested and then modified on the basis of the test results in order to improve it. There are systematic processes for evaluating solutions with respect to how well they meet the criteria and constraints of a problem. (secondary to MS‑PS3‑3)
*There is a strike through part of the DCI elements that are not developed in this unit. In the OpenSciEd Scope and Sequence, students will develop an understanding of changes in state, particularly as they relate to pressure, in OpenSciEd Unit 6.3, and frequency (color) of light in OpenSciEd Unit 8.4. The placement of this OpenSciEd Unit 6.2 and associated units are shown in the OpenSciEd Scope and Sequence.
Science & Engineering Practices

- Developing & Using Models
- Planning & Carrying Out Investigations
- Analyzing & Interpreting Data
- Constructing Explanations & Designing Solutions
- Engaging in Argument from Evidence
Crosscutting Concepts

- Systems & System Models
- Energy & Matter
- Structure & Function
Connections to the Nature of Science

Which elements of the Nature of Science are developed in this unit?
- Science investigations use a variety of methods and tools to make measurements and observations. (NOS-SEP)
- Science investigations are guided by a set of values to ensure accuracy of measurements, observations, and objectivity of findings.(NOS-SEP)
- Science depends on evaluating proposed explanations. (NOS-SEP)
- Science knowledge is based upon logical and conceptual connections between evidence and explanations (NOS-SEP)
- Science assumes that objects and events in natural systems occur in consistent patterns that are understandable through measurement and observation. (NOS-CCC)
- Science carefully considers and evaluates anomalies in data and evidence. (NOS-CCC)
How are they developed?
- Students engage with different tools (i.e., thermometers, light sensors, digital scales, simulations) to make measurements and observations.
- Students initially conduct uncontrolled investigations which prompts them to reconsider how they should better control their experiments. They also consider the accuracy of the data they collect from sampling errors and/or limitations of the tools they use.
- Students first investigate whether matter moving between systems explains the change in temperature. When they close the system and the temperature still changes, they consider whether light is causing the change in temperature. When a cup system placed in the dark still changes temperature, they begin to conduct a series of investigations on energy transfer via particle-scale mechanisms. The series described above engages students in collecting evidence to evaluate possible explanations for the water to cup up inside the cup.
- Students’ initial explanations about the cup warming up are not supported by evidence. Even though students cannot see the particles collide resulting in energy transfer, they can use macro scale experiences of collisions and computer simulations to make sense of what is happening between particles they cannot observe directly.
- Because students cannot observe particle motion directly, they must assume that collision between objects at the macroscale events share a similar pattern in energy transfer to at the particle scale, and that instruments we use, such as thermometers, can measure unobservable mechanisms.
- Students analyze data and determine what constitutes as an outlier (multiple lessons).
Unit Placement Information
What is the anchoring phenomenon and why was it chosen?

The anchoring phenomenon for this unit is a double-walled, plastic cup that looks similar to a regular plastic cup, but can keep a drink cold for longer than the regular plastic cup. These double-walled cups are often sold with claims that they can keep beverages colder (or warmer) for longer periods of time compared to other cups. Double-walled cups are made using air or vacuum insulation. This air- or vacuum-insulated layer minimizes energy transfer via conduction. The result is a cup that slows energy transfer allowing a cold drink to remain cold or a hot drink to retain its energy to remain warm for a longer period of time. Students will initially think they can explain the phenomenon using terms like ‘insulated’ or ‘vacuum sealed’ but when pressed further in Lesson 1, gaps in their understanding of how and why this works are revealed, which are used to motivate student learning throughout the unit.
The choice for a unit focus on double-walled cup designs was made for the following reasons:
- The disciplinary core ideas for energy transfer focus on transfer by conduction (i.e., particles colliding with each other). Thus, other energy transfer devices, such as a solar cooker or emergency blanket, while related to this anchoring phenomenon, would require knowledge of transfer by radiation that students have not yet developed in the middle school OpenSciEd Sequence. Thus, the unit needed a phenomenon that would allow students to explore conduction deeply. Radiation and convection are added to students’ understanding of energy transfer in the Storms Unit that follows this unit.
- This unit includes a substantial engineering component with multiple iterations on design. Designing any device in the classroom can be costly and material intensive, but designing cups allowed for fewer materials and an easier design process compared to other options.
- The phenomenon is pervasive in our lives. Cups of different materials are used at home, restaurants, and fast food chains. Expensive vacuum sealed cups and coolers tout their quality is worth the added price. This unit makes this everyday phenomenon puzzling to students by engaging them in explaining why certain materials and designs work by minimizing energy transfer. This unit intentionally makes the everyday experience ‘phenomenal’.
- A pre-field test of the pilot for a cup design anchor produced Driving Question Boards with productive students’ questions and ideas for investigations and sources of data needed to answer those questions. We used these early pilots of the anchor lesson and the field test feedback to anticipate the kinds of questions students would want to investigate throughout the unit. Teachers reported that students initially think they can explain the phenomenon but once they realize they do not understand vacuums and air insulations, there were high levels of engagement and curiosity about it.
- Focusing on keeping a drink cold, as opposed to keeping a drink warm, was preferred for safety reasons. Students participate in multiple labs using water or different temperatures. While they do work with warm water at times, we tried to limit use of warm or hot water. It was especially important in the design challenge to use cold water for safety, as opposed to hot water.
If students experience phenomenon fatigue with the cups given the length of the unit, it’s possible to broaden to related objects and devices, such as coolers, lunch boxes, or other devices that minimize energy transfer by slowing conduction. It is recommended not to broaden too far into energy transfer via convection or radiation as those ideas will be developed later in the OpenSciEd sequence.
How is the unit structured?


In the first lesson set, students conduct initial investigations into the cups to test cup features (components of the system) they conjecture may be important to keep the drink cold. They test their ideas with both cold and hot water to see how the cup features works in both situations. This motivates 1) the need for more systematic investigations to test the various cup features, and 2) wanting to look closely at the function of the lid (because initial data indicates that the lid is a crucial part of the system). Students design and carry out controlled investigations testing the lid. They figure out that the lid acts to close the system by trapping matter from escaping. They develop particle models to explain evaporation, mostly focused on modeling the structure of solids, liquids, and gases, and the movement of water particles from liquid to gas. Their models at this point are limited to explaining how particles of liquid gain energy and become gas particles moving into the air. Thus, they are left puzzled by the observation that when they completely close the system, the drink still warms up — even though they are sure that matter can no longer escape or enter the system from gaps in the lid area.
This limitation in their models motivates the second lesson set. In this lesson set, students investigate how energy can enter or leave the system. They investigate their ideas about light first, given they have more background about light from the previous One-way Mirror Unit. They investigate how light interacts with the cup walls and lid. Students figure out that the liquid inside cups with reflective walls warm up less than liquids inside cups with transparent and dark-colored walls. Using this evidence, they modify their models for explaining how light interacts with matter to include absorption of light. While these models explain more than their prior models, students find that, even when placed in a completely dark space, the liquid inside the cup warms up. This motivates the need for another mechanism of energy transfer. Students investigate particle motion in warmer and cooler substances via lab investigations and a series of simulations. Through these investigations, they collect evidence that suggests energy enters the cup system through particle collisions from the relatively warmer air outside the cup. Thus, they add this mechanism of energy transfer to their explanations of the phenomena so far.
In the third lesson set, students revisit popular and highly effective design features of cups, such as reflective materials and air insulation, and they model how these features minimize energy transfer by reducing absorption of light or minimizing the opportunity for particle collisions. They draw on their models to engage in a design challenge to design a cup that can compete with the expensive store-bought cups in keeping liquids cold. They use what they have figured out about mechanisms of energy transfer and design features that slow energy transfer so that the liquid in the cup keeps its original temperature longer. Students design, build, test, and modify their cup system through two design cycles. At the conclusion of this design challenge, students develop the ultimate cup design together, drawing upon features of successful cups. Then they apply their model for energy transfer to explain related phenomena they experience in their daily lives.
Where does this unit fall within the OpenSciEd Scope & Sequence?

This is the second unit in the 6th grade scope and sequence for OpenSciEd. There are six total units designed for 6th grade. This unit directly builds off the Disciplinary Core Ideas (DCIs), Crosscutting Concepts (CCCs), and Science and Engineering Practices (SEPs) developed in the first unit of 6th grade, the One-way Mirror Unit. The unit helps to complete coverage of element 1 of disciplinary core idea PS4.B Electromagnetic Radiation.
This unit is also the very first engineering unit for the OpenSciEd sequence and relies heavily on elementary engineering design Performance Expectations, while further developing engineering design focused on constraints, trade offs, and systematic testing and iteration.
The very next unit in the OpenSciEd Sequence, the Storms Unit will build off the DCIs, CCCs, and SEPs developed in this unit, further elaborating on particle models, matter transformation, and energy transfer in the context of micro to large-scale systems.
What modifications will I need to make if this unit is taught out of sequence?

This is the second unit in 6th grade in the OpenSciEd Middle School Scope and Sequence and it focuses on thermal energy. Given this placement, several modifications would need to be made if teaching this unit earlier or later in the middle school curriculum. These include:
- If taught before OpenSciEd Unit 6.1, supplemental teaching of light interactions with matter, such as reflection and transmission, would need to be added. These ideas are fundamental to the model students need to build of energy transfer through particle collisions.
- If taught before OpenSciEd Unit 6.1 or at the start of the school year, supplemental teaching of classroom norms, setting up the Driving Question Board, and asking open-ended and testable questions would need to be added. (These supports are built into 6.1.)
- If taught later in the OpenSciEd sequence after other engineering design challenges, modify the cup design challenge so that students are more involved in defining the problem and determining the criteria and constraints. Currently, Unit 6.2 is students’ first introduction to engineering design in the OpenSciEd 6–8th grade sequence. Therefore it is more scaffolded than it would need to be if the unit were taught much later in the middle school curriculum.
What are prerequisite math concepts necessary for the unit?

Beginning in Lesson 4 and throughout the unit, students focus on pooling and then averaging test results and building an understanding of temperature as a measure of average particle movement. They take measurements in the tenth or hundredth in decimal points, and must consider negative and positive numbers as they mass systems. Prerequisite math concepts that may be helpful include:
- CCSS.Math.Content.5.NBT.A.3 Read, write, and compare decimals to thousandths.
- CCSS.Math.Content.5.NBT.A.4 Use place value understanding to round decimals to any place.
- CCSS.Math.Content.6.SP.A.3 Recognize that a measure of center for a numerical data set summarizes all of its values with a single number, while a measure of variation describes how its values vary with a single number.
- CCSS.MATH.CONTENT.6.NS.C.5 Understand that positive and negative numbers are used together to describe quantities having opposite directions or values (e.g., temperature above/below zero, elevation above/below sea level, debits/credits, positive/negative electric charge); use positive and negative numbers to represent quantities in real-world contexts, explaining the meaning of 0 in each situation.
Calculating a mean of a data set is a target idea in 6th-grade CCMS. Prior to Lesson 4 in this unit, talk to your grade’s math teacher to find out when students will learn how to calculate the mean of a numerical data set in math class this year. If they have worked on this already, ask the math teacher for an example data set they worked with and any suggested modification to your anchor chart for calculating a mean. If students haven’t yet worked through any examples in their math classes, then the pooled temperature data in this lesson will be an example you save to refer to in future lessons in concert with this anchor chart. Also ask the math teacher if students have worked with using negative numbers to represent quantities in the real world. This will inform your decision about how to represent temperature changes in the pooled class data table.
How do I shorten or condense the unit if needed? How can I extend the unit if needed?

The following are example options to shorten or condense parts of the unit without eliminating important sensemaking for students:
- Eliminate or reduce Lesson 5 focused on condensation. Condensation on the outside of the cup may not become obvious to students and therefore not needed to be investigated. Condensation will be deeply explored in the next unit, Storms Unit.
- Shorten Lesson 7 to be a brief problematize move to motivate needing to investigate other ways to warm up the cup, focusing students on energy transfer.
- Instead of conducting the Lesson 9 lab, make a video of the lab in advance and watch it together as a class. Then discuss the patterns in temperature data.
- Eliminate the food coloring investigation in Lesson 10. The peppermint dye video is sufficient for developing the idea that particles is warmer liquids are moving faster.
- Sort the evidence from Lessons 11-13 as you go to eliminate the evidence sorting activity at the start of lesson 14. If students are fairly convinced that energy transfers from hotter regions to colder ones after Lesson 13, eliminate the butter demonstration, too.
- Eliminate the Lesson 15 posters of features that minimize energy transfer and gallery walk that follows.
To extend or enhance the unit, consider the following:
- Consider opening up the design challenge to other devices in which conduction is the primary means of energy transfer, such as coolers or lunch boxes.
- Involve students more in setting up the engineering design challenge to name their criteria and constraints and to identify how to test their devices against the criteria and constraints. Students could also interview stakeholders, such as other students, teachers, and family members to decide what criteria should be prioritized most.
Unit Acknowledgements
Unit Development Team

- Lindsey Mohan, Unit Lead, BSCS Science Learning
- Zoe Buck Bracey, Writer, BSCS Science Learning
- Emily Harris, Writer, BSCS Science Learning
- Ari Jamshidi, Writer, Stanford University
- Abe Lo, Writer, BSCS Science Learning
- Michael Novak, Writer & Reviewer, Northwestern University
- Tracey Ramirez, Writer, Charles A. Center at UT-Austin
- Dawn Novak, Pilot teacher, Maple School
- Tyler Scaletta, Pilot teacher, North Shore Country Day School
- Katie Van Horne, Assessment Specialist
- David Fortus, Unit Advisory Chair, Weizmann Institute of Science
Production Team

BSCS Science Learning
- Kate Herman, Copyeditor, Independent Contractor
- Stacey Luce, Editorial Production Lead
- Valerie Maltese, Marketing Specialist & Project Coordinator
- Alyssa Markle, Project Coordinator
- Chris Moraine, Multimedia Graphic Designer
Additional Acknowledgement

This unit was adapted from Energy: How can we design a device to warm something up?, originally co-developed by San Francisco Unified School District and SCALE at Stanford University 2017–2018. Used with permission. Energy: How can we design a device to warm something up? was developed with funding from Lucas Education Research, part of the George Lucas Education Foundation.
Unit External Evaluation
EdReports

EdReports awarded OpenSciEd an all-green rating for our Middle School Science Curriculum in February 2023. The materials received a green rating on all three qualifying gateways: Designed for the Next Generation Science Standards (NGSS), Coherence and Scope, and Usability. To learn more and read the report, visit the EdReports site.
NextGenScience’s Science Peer Review Panel

An integral component of OpenSciEd’s development process is external validation of alignment to the Next Generation Science Standards by NextGenScience’s Science Peer Review Panel using the EQuIP Rubric for Science. We are proud that this unit has earned the highest score available and has been awarded the NGSS Design Badge. You can find additional information about the EQuIP rubric and the peer review process at the nextgenscience.org website.

Unit standards
This unit builds toward the following NGSS Performance Expectations (PEs) as described in the OpenSciEd Scope & Sequence:
- MS-PS1-4*
- MS-PS3-3
- MS-PS3-4
- MS-PS3-5
- MS-PS4-2*
- MS-ETS1-4
Reference to kit materials
The OpenSciEd units are designed for hands-on learning and therefore materials are necessary to teach the unit. These materials can be purchased as science kits or assembled using the kit material list.